Enhanced photocatalysis by rGO-ZnO and rGO-Au-ZnO nanostructures
K Bramhaiah, V N Singh and Neena S John,
Phys. Chem. Chem. Phys. 2016, 18, 1478
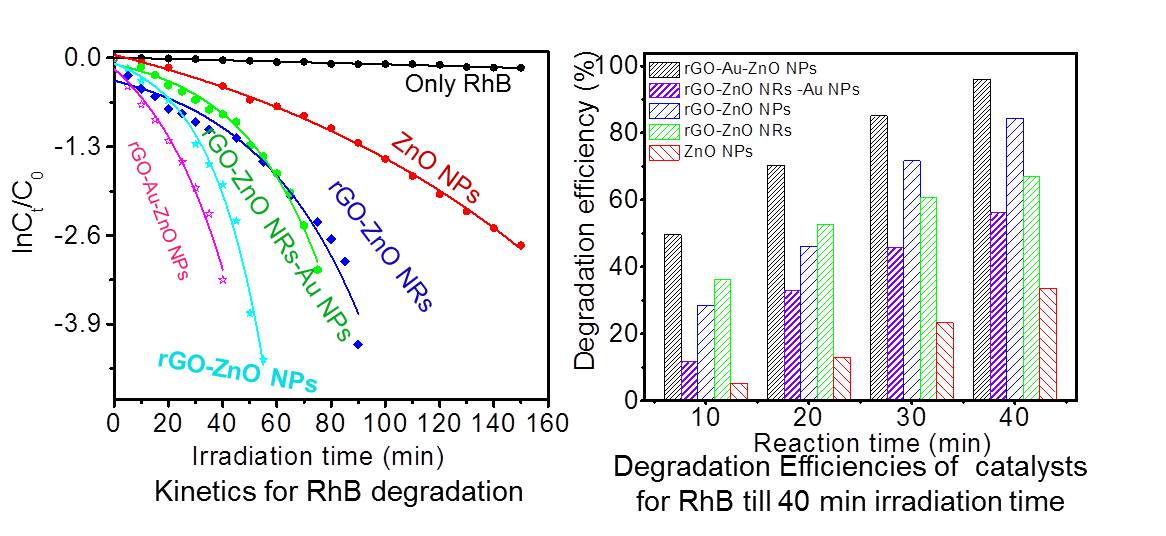
The influence of composition, morphology and defects on the photocatalytic activity of binary and ternary hybrid systems of rGO-ZnO and rGO-Au-ZnO are investigated. ZnO possessing various morphologies such as nanoparticles and nanorods on rGO and rGO-Au are obtained by solution phase and hydrothermal methods involving hydrolysis of zinc acetylacetonate in the presence of GO. Photocatalytic degradation studies of rhodamine B dye reveals that all the ZnO hybrids show higher degradation rates than bare ZnO. The photodegradation rates follow the order rGO-Au-ZnO NPs > rGO-ZnO NPs > rGO-Au-ZnO NRs> rGO-ZnO NRs > bare ZnO NPs. The difference in kinetics of ZnO hybrids is correlated with the observed photoluminescence, which is reflected as quenched band edge and defect emission arising from the charge transfer between rGO, ZnO, and Au leading to suppression in exciton recombination rates. Various factors such as structure, morphology, charge transfer and adsorption are considered to explain the observed kinetics.
